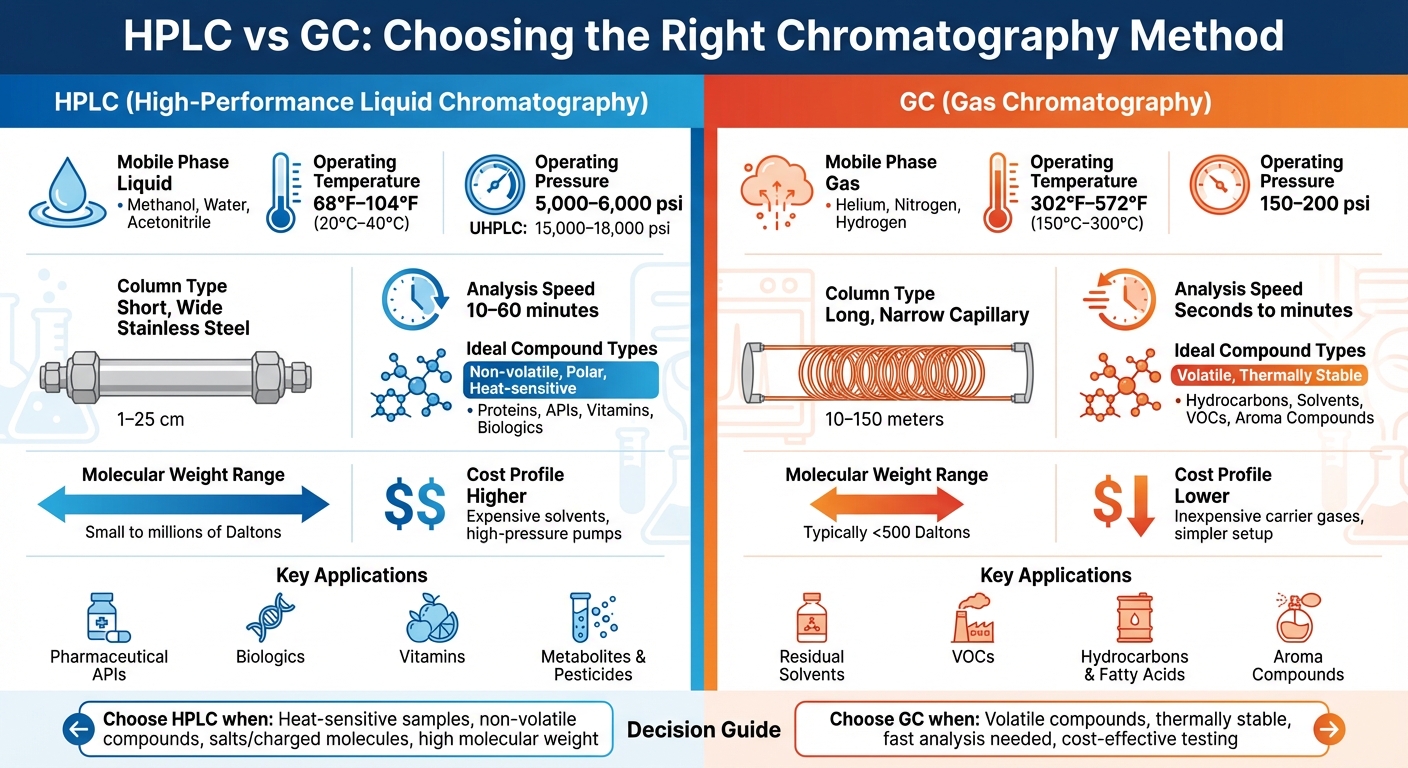
High-Performance Liquid Chromatography (HPLC) and Gas Chromatography (GC) are two widely used methods for analyzing chemical compounds, but they serve different purposes based on the properties of the sample. HPLC uses a liquid solvent to analyze non-volatile, polar, and heat-sensitive substances like proteins and vitamins. GC, on the other hand, uses a gas carrier to analyze volatile and heat-stable compounds such as hydrocarbons and solvents. The choice between these methods depends on factors like volatility, thermal stability, and the industry-specific application.
Key Differences:
- HPLC is ideal for non-volatile and heat-sensitive compounds, operating at lower temperatures (68°F–104°F). Common uses include pharmaceutical APIs, biologics, and vitamins.
- GC excels in analyzing volatile and thermally stable compounds, operating at high temperatures (302°F–572°F). Applications include residual solvents, aroma compounds, and hydrocarbons.
Quick Comparison:
| Feature | HPLC | GC |
|---|---|---|
| Mobile Phase | Liquid (e.g., methanol, water) | Gas (e.g., helium, nitrogen) |
| Operating Temperature | Ambient (68°F–104°F) | High (302°F–572°F) |
| Compound Type | Non-volatile, polar, heat-sensitive | Volatile, thermally stable |
| Speed | Moderate (10–60 minutes) | Fast (seconds to minutes) |
| Key Applications | APIs, vitamins, metabolites | Residual solvents, VOCs, hydrocarbons |
Selecting the right method ensures accurate results and prevents issues like sample degradation. Each technique plays a specialized role in industries such as pharmaceuticals, food safety, and petrochemicals. Always consider the sample’s properties and analytical goals when choosing between HPLC and GC.
HPLC vs GC Chromatography Methods Comparison Chart
Core Differences Between HPLC and GC
Mobile Phases and Operating Conditions
At the heart of the differences between High-Performance Liquid Chromatography (HPLC) and Gas Chromatography (GC) is the type of mobile phase each technique uses. HPLC employs liquid solvents like methanol, water, or acetonitrile to move samples through the column, while GC relies on inert gases such as helium, nitrogen, or hydrogen. This fundamental difference shapes the types of compounds each method can analyze.
"The mobile phase used in HPLC is a liquid whereas it is a gas in Gas Chromatography. Due to the higher density and viscosity of liquids a pump becomes necessary to drive the liquid through the HPLC column and as a result back pressure are high." – Dr. Deepak Bhanot, Analytical Chemistry Professional [2]
In HPLC, the liquid mobile phase interacts with analytes based on polarity and solubility, influencing how compounds separate. This method operates at room temperatures – typically around 68°F to 77°F (20°C to 25°C) – making it ideal for heat-sensitive substances like biologics, vitamins, and pharmaceutical actives that could degrade at higher temperatures. GC, on the other hand, uses its inert carrier gas to transport vaporized analytes without chemical interaction, with separation determined by volatility and boiling point. GC operates at much higher temperatures, ranging from 302°F to 572°F (150°C to 300°C), and can even reach 842°F (450°C) for specific applications, with operating pressures of around 150–200 psi [2][4].
These temperature differences also influence the pressure requirements of each system. HPLC systems rely on high-pressure pumps, capable of operating between 5,000 and 6,000 psi, with Ultra-High-Performance Liquid Chromatography (UHPLC) systems reaching pressures of 15,000 to 18,000 psi to push viscous liquids through tightly packed columns. This results in significant back pressure [2]. Such demands necessitate specialized designs for HPLC systems.
Equipment and Setup
The differences in mobile phases directly impact the equipment used in HPLC and GC. HPLC systems feature short, wide stainless-steel columns, typically ranging from 1 cm to 25 cm (0.4 inches to 10 inches) in length [2][7]. These columns are tightly packed, generating high back pressure, which requires the use of robust pumping systems. In contrast, GC systems utilize long, narrow capillary columns made from fused silica, extending anywhere from 10 to 150 meters (33 feet to 492 feet) [2][4]. These longer columns provide superior separation for volatile compounds while maintaining lower pressure requirements.
Another key distinction lies in the speed of analysis. GC, driven by high temperatures and the gaseous state of analytes, often completes separations in minutes or even seconds. HPLC, however, typically requires 10 to 60 minutes per run, partly due to the slower movement of liquids and the use of gradient elution in many applications [5][6]. Additionally, HPLC is the go-to method for samples containing salts or charged molecules that cannot be vaporized [8]. Understanding these equipment and operational differences is critical when selecting the best method for analyzing specific compounds.
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
HPLC and GC – what is the difference – animation HD
Analyte Types and Sample Compatibility
The effectiveness of HPLC or GC for compound analysis largely depends on the physical and chemical characteristics of the target analytes. Two key factors – volatility and thermal stability – play a decisive role in method selection. HPLC is better suited for compounds that are non-volatile or sensitive to heat, as these might degrade or fail to vaporize under high temperatures. On the other hand, GC works well for analytes that are volatile and stable enough to endure temperatures between 302°F and 842°F (150°C to 450°C) [2].
GC is typically used for smaller molecules under 500 Daltons, while HPLC can handle a broader range, including molecules as large as several million Daltons, such as proteins, polymers, and other biomolecules. Additionally, samples containing salts or electrically charged compounds are best analyzed with HPLC, as these cannot be vaporized for GC [8]. Below, we explore how each method addresses specific challenges associated with different analytes.
HPLC: Non-Volatile and Heat-Sensitive Compounds
HPLC operates at moderate temperatures (68°F–104°F), making it ideal for analyzing thermally unstable compounds. In the pharmaceutical sector, HPLC is essential for quantifying active pharmaceutical ingredients (APIs), analyzing biologics like proteins and amino acids, and detecting metabolites in complex biological samples. Similarly, the food and beverage industry uses HPLC to measure vitamins, mycotoxins, and additives that could degrade at the high temperatures required for GC.
"HPLC is particularly well-suited for non-volatile, polar, thermally unstable, and high-molecular-weight compounds, making it indispensable in pharmaceutical analysis, clinical research, food and beverage testing, and environmental monitoring." – Trevor J Henderson, Lab Manager [1]
Environmental laboratories rely on HPLC to analyze pesticides, herbicides, and pharmaceutical residues in water and soil. The petrochemical industry uses it to study polymers, degradation products, and industrial additives. HPLC also plays a key role in analyzing explosive materials, as it avoids the safety risks and complications associated with the high temperatures needed for GC. While HPLC excels in these areas, GC is better for volatile compounds, as discussed below.
GC: Volatile and Heat-Stable Compounds
GC is ideal for analyzing volatile organic compounds (VOCs) and substances that can form stable vapors when heated. This method is widely used in industries examining hydrocarbons, refinery gases, fuels, and lubricants. Environmental testing labs use GC to measure air pollutants, VOCs, and semi-volatile organics in soil and water. The food industry employs GC to analyze aroma compounds, flavor volatiles, and fatty acids, which are critical for assessing product quality.
"Gas chromatography is the method of choice for analyzing mixtures made up of anything that can be heated to form stable vapors." – Tara M. Lovestead, Applied Chemicals and Materials Division, NIST [4]
Pharmaceutical manufacturers use GC to detect residual solvents and volatile organic impurities in drug formulations, ensuring compliance with regulatory standards. Forensic laboratories also depend on GC for identifying toxins, alcohol levels, and volatile poisons in biological samples. With detection limits as low as 0.5 pg, GC-MS offers exceptional sensitivity for trace-level analysis. Additionally, GC separations are significantly faster, often taking just seconds or minutes, compared to the 10- to 60-minute runs typical for HPLC [5].
Comparison Table: Analyte Types and Applications
| Industry | HPLC Applications | GC Applications |
|---|---|---|
| Pharmaceuticals | API quantification, biologics, metabolites [1] | Residual solvents, volatile impurities [1] |
| Food & Beverage | Vitamins, mycotoxins, additives [1] | Aroma compounds, flavor volatiles, fatty acids [1] |
| Environmental | Pesticides, herbicides, pharmaceutical residues [1] | VOCs, air pollutants, semi-volatile organics [1] |
| Petrochemical | Polymers, degradation products, additives [1] | Hydrocarbons, refinery gases, fuels, lubricants [1] |
This content is for informational purposes only. Consult official regulations and qualified professionals before making sourcing or formulation decisions.
sbb-itb-aa4586a
Applications in Industrial Testing and Quality Control
HPLC and GC play essential roles in maintaining industrial quality standards by focusing on different compound characteristics. Each technique is suited for specific applications, and understanding their differences allows laboratories to choose the most effective method for their needs. Below is an overview of how these methods cater to various industrial requirements.
HPLC Applications
In pharmaceutical and clinical industries, HPLC is the gold standard for quantifying active pharmaceutical ingredients (APIs), monitoring product stability, and profiling impurities. It operates at ambient temperatures (68°F–104°F/20°C–40°C), which is crucial for preserving delicate compounds like proteins and metabolites that might degrade under high heat. Clinical and forensic labs also rely on HPLC to detect drugs of abuse and their metabolites in biological samples such as blood and urine, where its ability to analyze charged compounds and salts is indispensable.
In the food and beverage industry, HPLC is widely used to measure heat-sensitive vitamins (like B-complex, C, A, D, E, and K), food additives such as preservatives and artificial sweeteners, and toxins like aflatoxin B1 and domoic acid. Environmental laboratories also turn to HPLC for detecting non-volatile pollutants. Beyond these areas, HPLC supports industrial and materials science by characterizing synthetic polymers, additives, and degradation products.
GC Applications
On the other hand, GC excels in applications requiring high-temperature analysis and is a staple in industries like petrochemical and oil & gas. It is used to analyze refinery gases, crude oil components (e.g., aromatics and BTEX), fuels, and lubricants. With operating temperatures ranging from 302°F to 572°F (150°C–300°C), GC is perfect for vaporizing and separating volatile compounds. Environmental labs also use GC to measure volatile organic compounds (VOCs) and air pollutants, with GC-MS systems capable of analyzing samples containing hundreds of compounds [9].
The food and fragrance industries depend on GC for profiling essential oils, aroma compounds, flavor volatiles, and fatty acids, all crucial for quality control.
"GC is the method of choice for any mixture that can be heated to form stable vapors, offering higher resolution for complex volatile mixtures due to the high efficiency of long capillary columns." [4]
Forensic and toxicology labs rely on GC to detect toxins and drugs of abuse in various sample types. In pharmaceuticals, GC is employed to identify residual solvents and volatile impurities in formulations, ensuring compliance with regulatory standards. Its speed is a major advantage, with analyses often completed in seconds to minutes, compared to HPLC’s 10- to 60-minute run times [5].
Comparison Table: Speed and Matrix Complexity
| Feature | HPLC | GC |
|---|---|---|
| Analysis Speed | Moderate (10–60 minutes) [5] | Fast (seconds to minutes) [5] |
| Matrix Compatibility | Excellent for liquid, biological, and food matrices [1] | Best for gaseous, volatile, and semi-volatile mixtures [4] |
| Precision/Resolution | High; sharp symmetrical peaks [1] | Very high; narrower, sharper peaks for volatiles [4] |
| Operating Temperature | Ambient (68°F–104°F / 20°C–40°C) [5] | High (302°F–572°F / 150°C–300°C) [5] |
| Sample Preparation | Filtration and degassing; handles complex matrices | Often requires extraction from "dirty" matrices |
This information is provided for general guidance. Always consult official standards and experts when making decisions about sourcing or formulations.
Cost and Selection Factors for Chromatographic Methods
When deciding on a chromatographic method, factors like cost and regulatory requirements are just as critical as operating conditions and sample compatibility.
Equipment and Operating Costs
The expenses associated with High-Performance Liquid Chromatography (HPLC) and Gas Chromatography (GC) go well beyond the initial purchase price. HPLC systems, for instance, demand high-pressure pumps, intricate injectors, and specialized columns. These components contribute to higher upfront costs. Additionally, the ongoing use of high-purity solvents like acetonitrile and methanol, along with their disposal, adds significantly to operational expenses [1][5].
"HPLC tends to be more expensive than GC due to the need for high-pressure pumps, costly solvents, specialized columns, and a more complex, maintenance-intensive setup."
– Duilio Romanello [5]
On the other hand, GC is often a more budget-friendly option for many labs. Its initial equipment costs are lower, and operational expenses are kept in check because it uses inexpensive inert carrier gases like nitrogen or helium instead of costly solvents [1]. GC systems also require less maintenance due to their simpler gas delivery setup, though the columns may need more frequent replacement due to thermal stress from operating temperatures ranging from 302°F to 572°F (150°C–300°C) [5]. Another advantage of GC is its speed – analyses are completed in seconds to minutes, compared to HPLC’s longer run times of 10 to 60 minutes. This faster throughput directly translates to lower costs per analysis [5].
"Gas chromatography simply requires gas containers and carrier gas, which is more affordable than solvents. As a result, GC is generally seen as the more cost-effective option."
– Brewer Science [8]
These cost considerations also play a role in meeting compliance requirements in regulated environments.
Regulatory Compliance and Industry Standards
Regulatory standards are another key factor influencing the choice between HPLC and GC. In industries like pharmaceuticals, method selection often hinges on the chemical properties of the analytes and the regulatory requirements. HPLC is the go-to technique for quantifying active pharmaceutical ingredients (APIs), conducting stability studies, and profiling impurities. This is because most drugs are non-volatile or may degrade at the elevated temperatures required for GC [1]. However, for testing residual solvents or volatile organic impurities, regulatory guidelines specifically call for GC analysis [1].
The food industry follows a similar pattern. HPLC is commonly used for measuring compounds like vitamins, additives, and mycotoxins – substances that are sensitive to heat. Meanwhile, GC is favored for applications such as pesticide residue analysis and fatty acid profiling [3]. Both methods are designed to meet the stringent requirements of standards like the United States Pharmacopeia (USP) and Food Chemicals Codex (FCC), ensuring the precision needed to detect trace impurities [8]. Rigorous quality assurance protocols, including the use of reagent blanks and mixed standards, are also implemented to uphold system performance and data reliability [10].
Pros and Cons Table: HPLC vs GC
The table below highlights the key strengths and limitations of HPLC and GC to assist in method selection.
| Factor | HPLC | GC |
|---|---|---|
| Cost | Higher equipment and solvent costs; longer column lifespan | Lower initial investment; minimal solvent use; may require more frequent column replacement |
| Speed | Slower analysis (10–60 minutes) | Faster analysis (seconds to minutes) |
| Sample Type | Suitable for non-volatile, polar, and heat-sensitive compounds (covers about 80% of compounds) | Best for volatile, thermally stable compounds; polar compounds may need derivatization |
| Maintenance | More maintenance-intensive due to complex pump systems | Lower maintenance needs thanks to simpler gas delivery systems |
| Regulatory Use | Preferred for APIs, biologics, and vitamins | Required for residual solvents and volatile organic compounds analysis |
This table provides general guidance. Always consult official standards and experts for specific decisions regarding sourcing or formulations.
Choosing the Right Method for Your Needs
Deciding between HPLC and GC depends on the properties of your sample and its intended application. Start by evaluating your sample’s volatility and thermal stability. For volatile, thermally stable compounds – like hydrocarbons, residual solvents, or aroma compounds – that can endure temperatures up to 842°F (450°C), GC is often the go-to choice. On the other hand, if you’re working with non-volatile, polar, or heat-sensitive substances, such as proteins, biologics, or most pharmaceutical APIs, HPLC is usually the better option [1].
Molecular weight is another critical factor. GC is best suited for analyzing compounds with molecular weights in the low hundreds, whereas HPLC can handle a broad spectrum – from small molecules to large polymers and biomolecules with molecular weights in the millions. Additionally, samples containing salts or charged compounds are incompatible with GC’s vaporization process, making HPLC the necessary choice [1].
Practical considerations also play a role in method selection. GC offers faster analysis, often completing runs in seconds to minutes, which is ideal for high-throughput applications like petrochemical or environmental testing. HPLC, in contrast, requires 10 to 60 minutes per run, but it excels in analyzing complex biological matrices where analytes are polar or ionic [5]. Cost is another factor: GC tends to be more economical due to the use of inexpensive carrier gases like nitrogen or helium. HPLC, however, involves higher costs for high-purity solvents and high-pressure pumps, which contribute to both initial and ongoing expenses [5].
Regulatory standards also influence the choice of method. In the pharmaceutical industry, HPLC is required for API quantification and impurity profiling, while GC is used for analyzing residual solvents and volatile compounds [1]. Many labs utilize both techniques to meet diverse testing needs. For instance, in food safety, HPLC might measure vitamins and additives, while GC handles pesticide residues and fatty acids [3].
For industries like pharmaceuticals and food production, sourcing high-quality materials is equally important. Allan Chemical Corporation (https://allanchems.com) provides technical-grade and compendial-grade solutions tailored to these regulated sectors, backed by over 40 years of expertise and just-in-time delivery. Choosing the right analytical method goes hand-in-hand with ensuring reliable sourcing to meet industry standards.
This content is for informational purposes only. Always consult official regulations and qualified professionals before making sourcing or formulation decisions.
FAQs
What are the key factors to consider when choosing between HPLC and GC for compound analysis?
When choosing between high-performance liquid chromatography (HPLC) and gas chromatography (GC), the decision largely hinges on your sample’s characteristics and the specific analysis needs. GC works best for volatile compounds, substances with low molecular weight, and analytes that can withstand high temperatures. On the other hand, HPLC is better suited for thermally sensitive compounds, higher molecular weight substances, or polar analytes that dissolve in liquid solvents.
The type of mobile phase is another key consideration. HPLC uses liquid solvents selected based on the sample’s polarity and solubility, while GC employs inert gases like helium or nitrogen to transport the analytes. Temperature requirements also differ significantly: HPLC operates at room temperature or slightly elevated conditions, making it ideal for heat-sensitive samples. In contrast, GC requires higher temperatures to vaporize the compounds for analysis.
For labs across the United States, Allan Chemical Corporation offers high-purity carrier gases, HPLC solvents, and specialty columns. These products ensure dependable performance for industries such as pharmaceuticals, food, cosmetics, and electronics, where precision and reliability are crucial.
What are the cost differences between operating HPLC and GC systems?
While the article doesn’t dive into exact cost comparisons between operating HPLC (High-Performance Liquid Chromatography) and GC (Gas Chromatography), it’s clear that each method comes with its own set of expenses. These costs can stem from factors like equipment maintenance, consumables, and energy consumption. To get a precise understanding of the financial implications, it’s important to assess these elements in the context of your specific application and industry requirements.
What are the regulatory considerations when choosing HPLC or GC in pharmaceutical analysis?
In the pharmaceutical world, deciding between HPLC (High-Performance Liquid Chromatography) and GC (Gas Chromatography) carries specific regulatory requirements due to their unique applications. HPLC is commonly used for analyzing non-volatile substances, such as active pharmaceutical ingredients (APIs), impurities, and degradation products. Compliance with ICH Q2(R1) and FDA guidelines is essential for method validation, which includes ensuring factors like accuracy, precision, specificity, linearity, and sensitivity (limits of detection and quantitation). Regulatory authorities also require detailed method-development documentation and routine system suitability checks to guarantee consistent and reliable results.
GC, by contrast, is the go-to method for volatile or thermally stable compounds, such as residual solvents and hydrocarbons. Regulatory submissions for GC must adhere to standards like USP <467> or ICH Q3C, which outline residual solvent limits. Validation efforts focus on ensuring temperature stability, carrier gas purity, and detector linearity, all of which are critical for maintaining analyte stability under high-temperature conditions.
Allan Chemical Corporation supports these stringent regulatory demands by offering high-quality USP-grade solvents, chromatography columns, and reference standards. These products are designed to ensure dependable compliance for both HPLC and GC methodologies.





Comments are closed